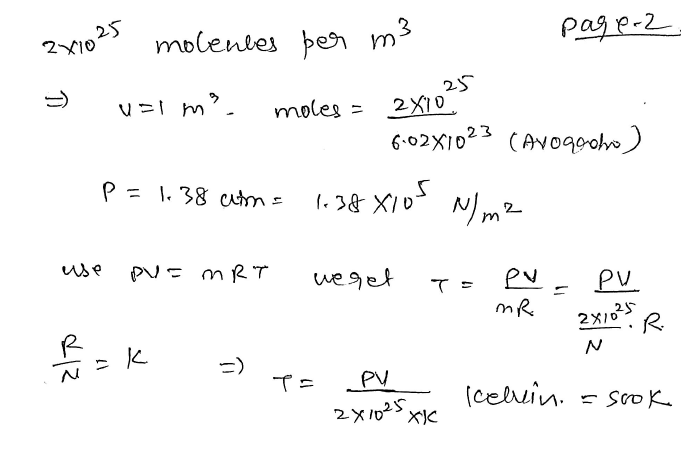Article
The temperature of a gas having 2.0 × 10^25 molecules per cubic meter at 1.38 atm (Given, k = 1.38 × 10^–23 Joule per Kelvin) is: JEE 2024
Volume is not given so assume it to be 1 meter cube then corresponding moles(n) can be calculated using the number density given in question so that we can use ideal gas equation.