
Books by Author Lovekush Kumar
Explore the books by author for excelling in your discipline.
Read NowAppreciation and Promotion
Explore for "promotable images/quotes" at your social media for a better society.
Read Now

Recent Articles
Change of temperature of a body in degree centigrade and kelvin
It has been observed that many students commit a mistake to add 273 in temperature difference while converting the temperature difference for a body in degree centigrade to kelvin. While is to be noted that if temeperature of a body is changed by t degree at centigrade scale then the temperature change at kelvin scale will be just t kelvin not t + 273.
Mathematically
t = t2 - t1 degree centigrade
difference at kelvin scale will be t2 + 273 - (t1 + 273) = t2 - t1
both have same numerical value.
Principle of continuity and its analogy with watering of garden
In the chapter for properties of liquid the principle of continuity states that velocity will be greater where cross sectional area of the pipe is smaller and vice-vers, the same can be remembered with analogy of watering pipe, when we pinch its opening a little bit water comes out of it with greater velocity and goes to much larger distance helping us to cover larger area by standing at a single location.
Calorimetry and change of state
Heat is energy in transition, from higher temperature body to body at lower temperature.
If "no loss of energy" then energy absobed by cooler body = energy exhausted by hotter body
For example if m gram ice cube is droped in M gram hot water then to calculate the final temperature of the mixture following step by step method may be employed:
if value of M and its temperature is sufficient enough to melt the ice
then heat required to melt the ice is mL where L is latent heat of fusion for ice.
heat required to increase the temperature of this m gram 0 degree water is ms(T - 0) where s is the specific heat for water and T is the final temperature of the mixture.
Heat exchausted by M gram hot water during cooling down from T1 to T is MS(T1 - T).
as per calorimetry MS(T1 - T) = mL + msT
Note- First ice will fully melt only then its temperature will start increasing provided energy is availble with hot water in other words it is hot enough.
In a series LCR circuit, the inductance L is 10 mH, capacitance C is 1 micro Farad and resistance R is 100 ohm. The frequency at which resonance occurs is? NEET UG2023

The magnetic energy stored in an inductor of inductance 4 micro-H carrying a current of 2 A is? NEET UG2023

The angular acceleration of a body, moving along the circumference of a circle, is DIRECTED along? NEET UG2023

The ratio of frequencies of fundamental harmonic produced by an open pipe to that of closed pipe having the same length is? NEET UG2023

A football player is moving southward and suddenly turns eastward with the same speed to avoid an opponent. The force that acts on the player while turning is? NEET UG2023
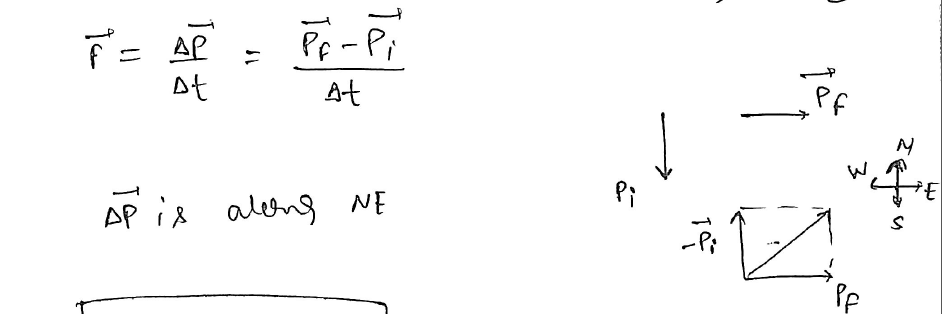
The temperature of a gas is –50°C. To what temperature the gas should be heated so that the rms speed is increased by 3 times? NEET UG2023
Note1- increased to 3 times and increased by 3 times are different.
In former case final value is 3 times while in latter case the final value is (3+1=4)times of the initial value.
Note-2 the formula has temperature in kelvin so first convert given value to kelvin.

